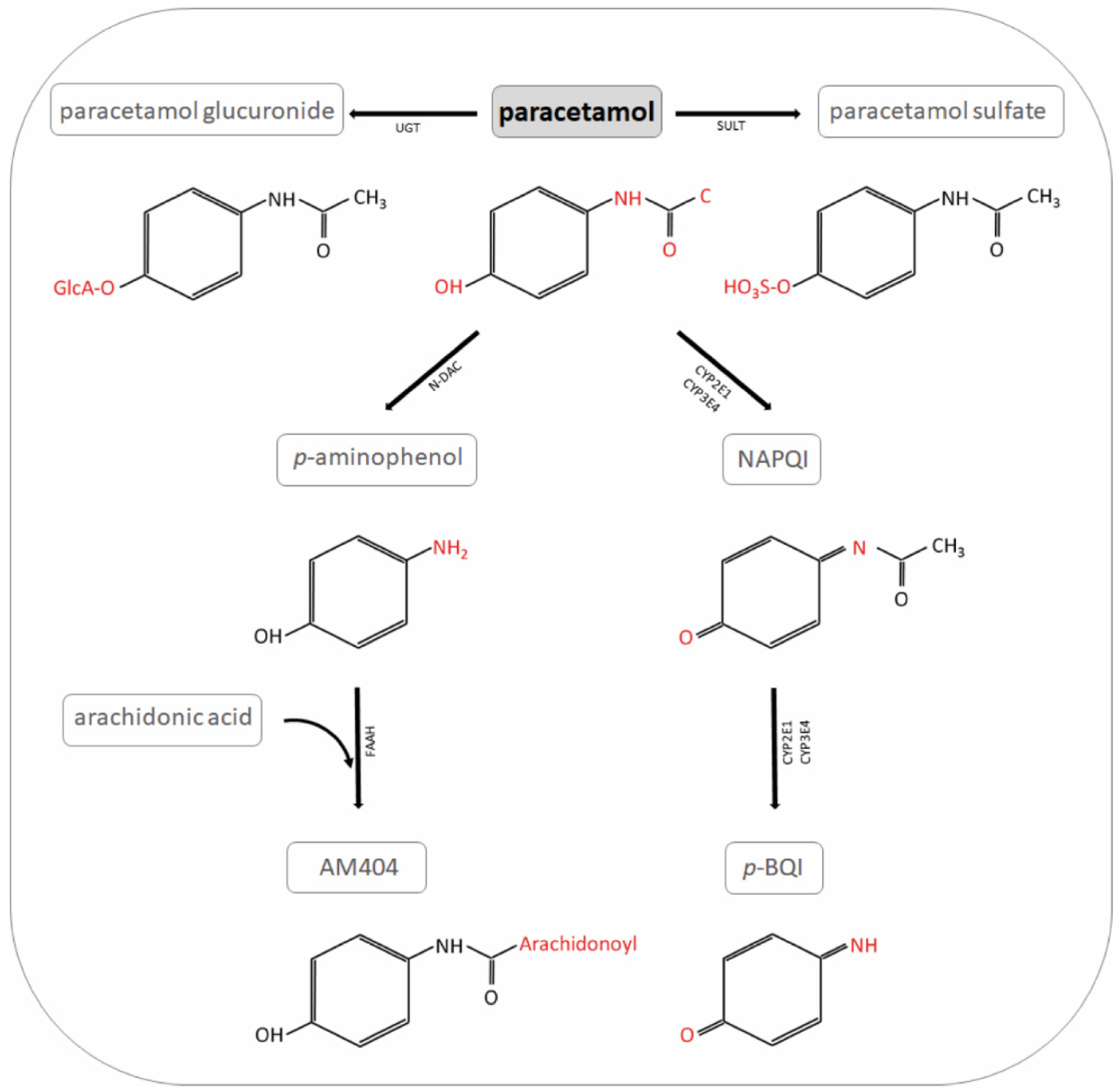
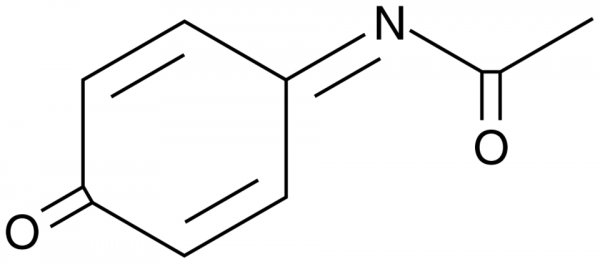
Tunable oxidative release of N-acetyl-p-benzoquinone-imine and acetamide from electrochemically derived sub-monolayer acetaminophen modified glassy carbon electrode - ScienceDirect

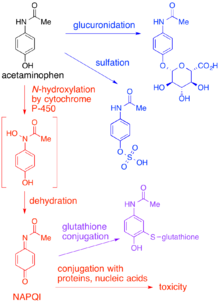
N-acetyl-p-benzoquinone imine: a cytochrome P-450-mediated oxidation product of acetaminophen. | Semantic Scholar

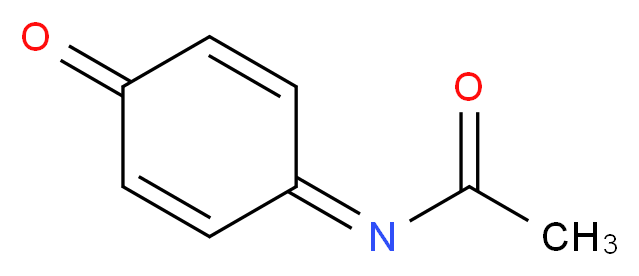
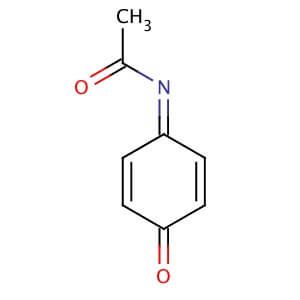
Synthesis of N-acetyl-p-benzoquinone imine (NAPQI) from acetaminophen. | Download Scientific Diagram

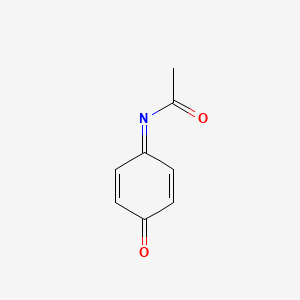
What Are the Potential Sites of Protein Arylation by N-Acetyl-p-benzoquinone Imine (NAPQI)? | Chemical Research in Toxicology

What Are the Potential Sites of Protein Arylation by N-Acetyl-p-benzoquinone Imine (NAPQI)? | Chemical Research in Toxicology
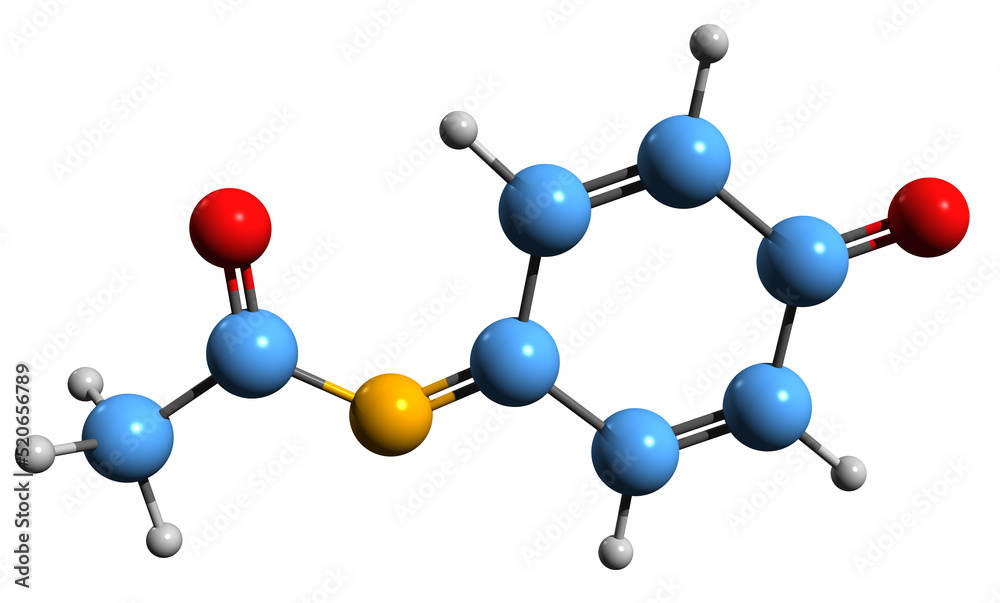
3D image of NAPBQI skeletal formula - molecular chemical structure of N- acetyl-p-benzoquinone imine isolated on white background Stock Illustration | Adobe Stock

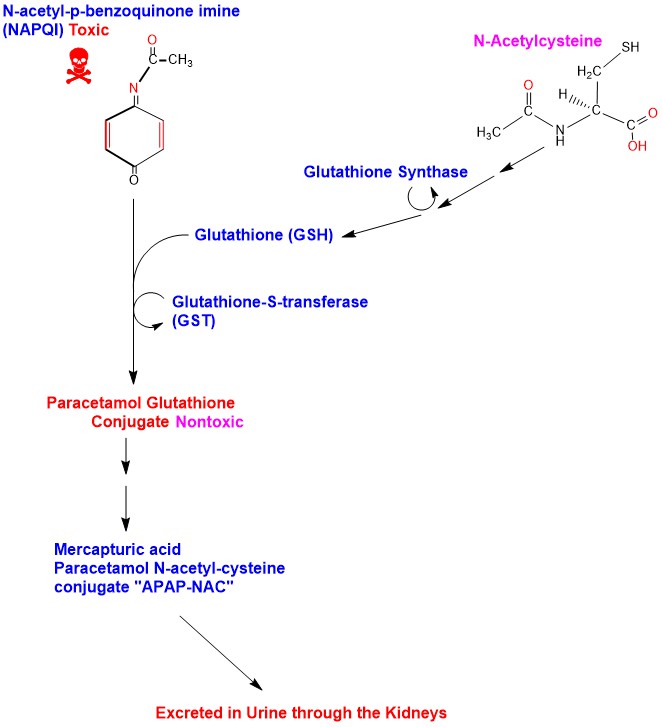
N-Acetyl-p-benzoquinone Imine, the Toxic Metabolite of Acetaminophen, Is a Topoisomerase II Poison | Biochemistry

![N-Acetyl-P-benzoquinone imine dimethyl ketal - Optional[13C NMR] - Chemical Shifts - SpectraBase N-Acetyl-P-benzoquinone imine dimethyl ketal - Optional[13C NMR] - Chemical Shifts - SpectraBase](https://spectrabase.com/api/spectrum/2qv4VDfwlbm/structure.png?h=300&w=382)